Research
What we do:
Major depression is a leading cause of disability worldwide, and many people do not benefit from current treatments. To develop better treatments, we need to understand how the neural circuits that control emotional behaviors are altered in disease. We also need to precisely map the circuits in order to modulate them with maximum therapeutic benefit and minimal side effects. We use innovative neuroscience approaches to tackle these challenges.
Alzheimer’s disease causes devastating memory impairments. These core symptoms of Alzheimer’s disease are due to a breakdown in neural computation, likely in the entorhinal cortex–hippocampal circuit. Successful treatments must address this computational breakdown. However, it is unknown how computation deteriorates during learning and memory in Alzheimer’s disease. We know many of the molecular-cellular drivers of disease, and we know the ruinous consequences for people’s lives, but the process connecting these – entorhinal-hippocampal computation – is poorly understood. Using mouse models, we determine how neural computation breaks down, and we develop methods to reverse this.
How we do it:
To determine how circuit activity breaks down in these diseases, we start with robust findings in humans (e.g. genetic risk factors, pharmacology) and we model these in mice. We use several in vivo imaging tools (two-photon, miniature microscopy, and fiber photometry) to record neural activity during behaviors most relevant to the human disease (emotional behaviors, learning and memory, sleep and circadian rhythms). We build computational models based on these recordings to determine what changes in activity need to be restored to reverse the disease states. To map the anatomy of the circuits involved, and how they change in disease, we use light sheet imaging of clarified brains. We use optogenetics and DREADDs to test new approaches to restore circuit function with high precision.
Our projects:
Computational breakdown in Alzheimer’s disease
We developed an integrated approach to measure behavior (learning and memory, sleep), record neural activity, and model neural computation in mouse models of Alzheimer’s disease. We use in vivo circuit manipulation to reverse the deficits we discover and to explore new treatment approaches.
Neural mechanisms of rare depression-associated mutations
We and our collaborators identified rare mutations in G-protein-coupled receptors associated with major depression in humans. We are now determining how these mutations change brain activity and drive symptoms. To do so, we model the mutations in mice and use in vivo imaging to record neural activity during emotional behavior. To develop new treatments, we test pharmacological and activity-based interventions to reverse these changes in brain activity.
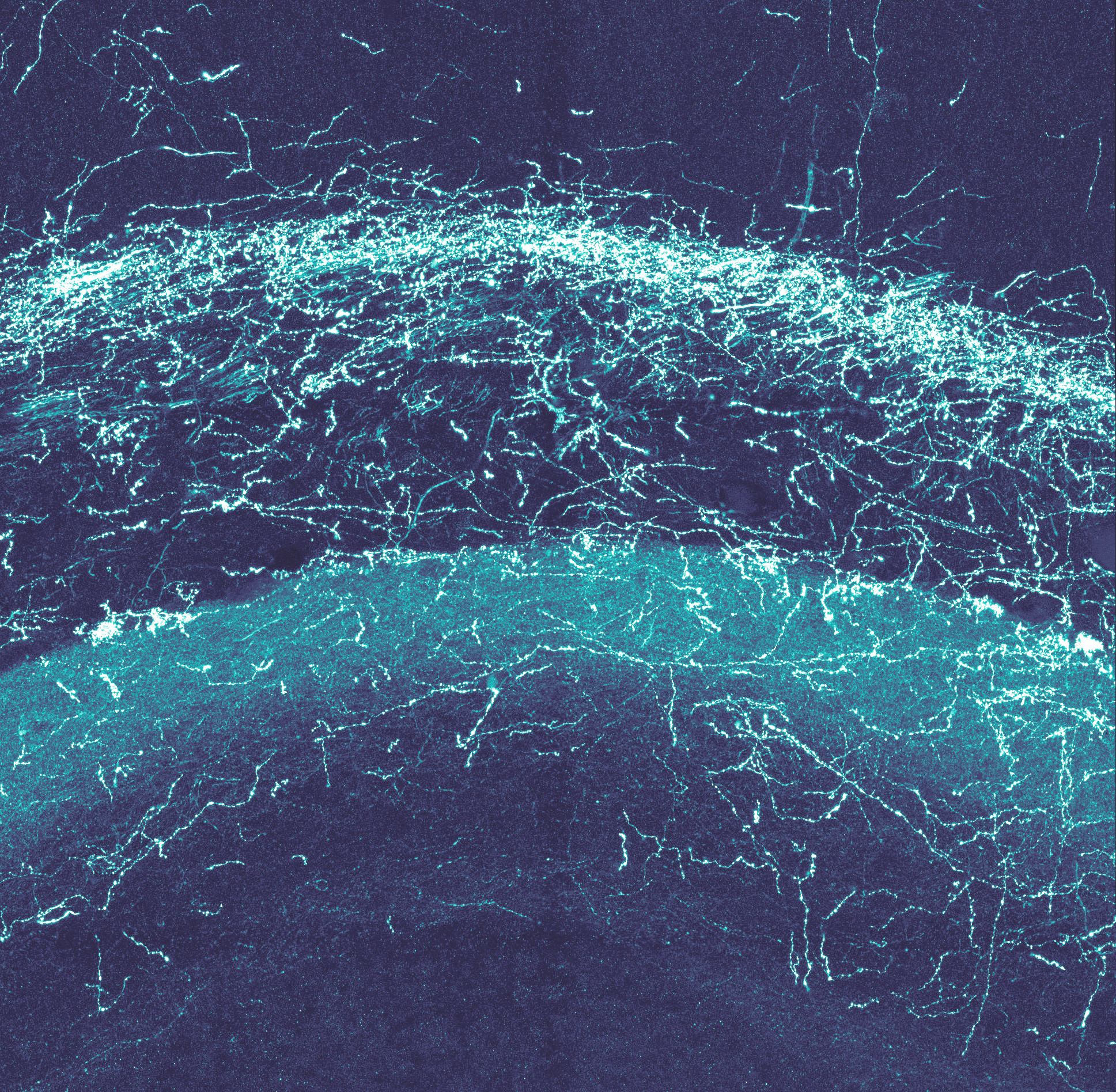
Functional mapping of serotonergic circuits
Using in vivo imaging, we found that serotonergic neurons are strongly activated by emotionally intense stimuli (both pleasurable and painful). We are now mapping how these serotonergic signals are broadcast throughout the brain. To do so, we use innovative functional and anatomical mapping across the mouse brain. Our goal is to build a fine-grained functional map of the serotonin system to develop more precise and effective treatments for depression.